 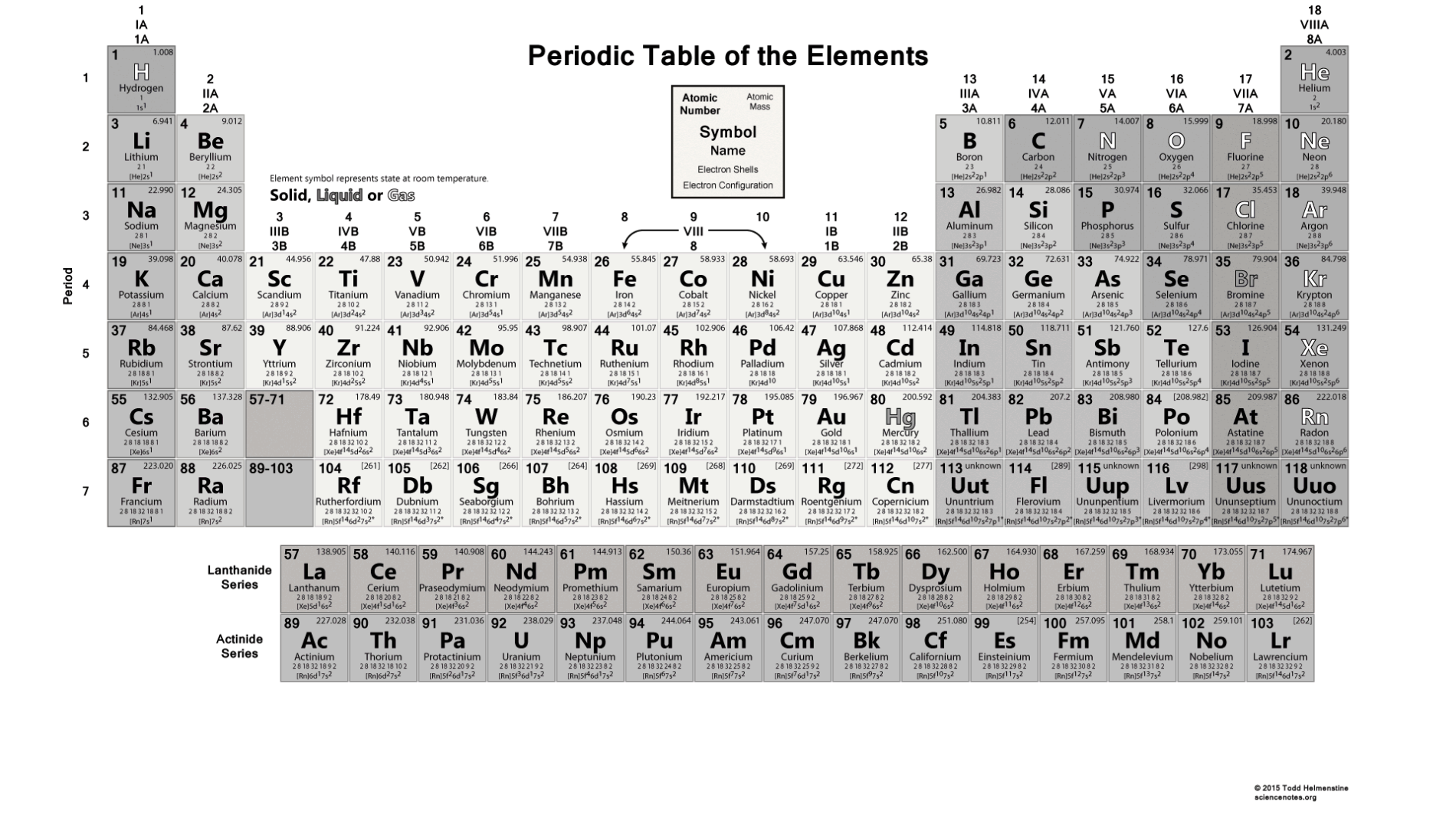
It also helps to know about the elements in the group. Periodic Table of Elements with Names and Symbols PDFĭo you know there are many advantages of the Periodic Table of Elements with Names and Symbols PDF? In the periodic table, you will find the atomic number of the elements and some fundamental properties of the elements. If you are comfortable with your device, you can also use the softcopy of the table to know the elements more. We suggest you download the bigger one if you are going to print it out of the table. We have a periodic table in the landscape as well as a portrait version. If you are a new learner or want to learn the table for the first time, you should download the basic periodic table. if you are working on particular properties, we also have a separate Periodic Table With Names And Atomic for every property. These tables include the information of elements like atomic mass, atomic number, electronegativity, ground-state level, atomic radius, etc. 
Sometimes geologist also needs this to know the elements found on the surface. The periodic table is necessary for all students, chemists as well as scientists also. The elements in the table are analyzed in a manner so that anyone can easily know the properties of the elements. This fact has key implications for the building up of the periodic table of elements.Are you searching for the Periodic Table With Names, Atomic Mass and Number? The periodic table is also famous as the periodic table of elements. The ordering of the electrons in the ground state of multielectron atoms, starts with the lowest energy state (ground state) and moves progressively from there up the energy scale until each of the atom’s electrons has been assigned a unique set of quantum numbers. It is the Pauli exclusion principle that requires the electrons in an atom to occupy different energy levels instead of them all condensing in the ground state. Each electron is influenced by the electric fields produced by the positive nuclear charge and the other (Z – 1) negative electrons in the atom. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. The total number of protons in the nucleus of an atom is called the atomic number (or the proton number) of the atom and is given the symbol Z. In the periodic table, the elements are listed in order of increasing atomic number Z. The number of electrons in each element’s electron shells, particularly the outermost valence shell, is the primary factor in determining its chemical bonding behavior. The configuration of these electrons follows from the principles of quantum mechanics. The chemical properties of the atom are determined by the number of protons, in fact, by number and arrangement of electrons. There is a recurring pattern called the “periodic law” in their properties, in which elements in the same column (group) have similar properties. Generally, within one row (period) the elements are metals to the left, and non-metals to the right, with the elements having similar chemical behaviours placed in the same column.Įvery solid, liquid, gas, and plasma is composed of neutral or ionized atoms. It is organized in order of increasing atomic number. The periodic table is a tabular arrangement of the chemical elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
